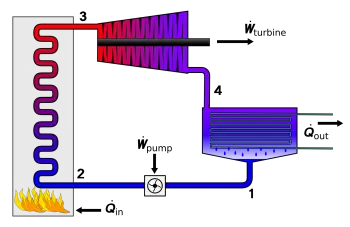
Rankine cycle. The conversion of heat into work and its uses
The Rankine cycle is a thermodynamic cycle composed of two isentropic transformations and two isobars. Its purpose is to transform heat into work using a heat exchanger. It is the basis of the design of steam engines of any type.
In an isentropic process the entropy of the system remains constant while in an isobaric process what remains constant is the pressure.
The Rankine cycle can be:
-
Open Rankine cycle, with the discharge of steam into the atmosphere (as for example, the old steam locomotives).
-
Closed Rankine cycle, as in the case of thermoelectric plants. In closed cycles, the residual heat from steam condensation can be used through cogeneration.
How does the Rankine cycle work?
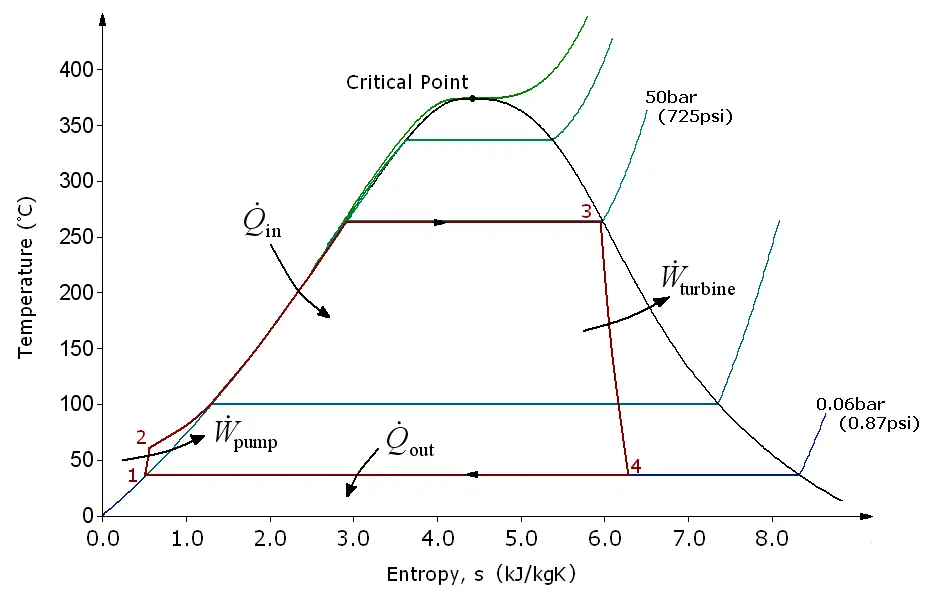
The Rankine cycle works with four thermodynamic processes. The states are identified by numbers (in brown) on the T - S diagram (temperature - entropy.)
-
Process 1-2: isentropic compression in the pump. The working fluid is pumped from low to high pressure. Since the fluid is a liquid at this stage, the pump requires little input energy.
-
Process 2-3: adding heat at constant pressure in the boiler. The high pressure liquid enters a boiler, where it is heated at constant pressure through an isobaric process by an external heat source to become a saturated dry steam. In this phase, a phase change of the water from liquid to gas takes place.
-
Process 3-4: isentropic expansion in turbine. The saturated dry steam expands through a steam turbine, generating energy. From a thermodynamic point of view, this lowers the temperature and pressure of the vapor, and some condensation can occur.
-
Process 4-1: constant pressure heat rejection in the condenser, isobaric process. The wet steam enters a condenser, where it condenses at a constant pressure to become a saturated liquid.
In an ideal Rankine cycle, the pump and turbine would be isentropic. That is, the pump and turbine would not generate entropy and therefore it will maximize the net work output. Processes 1-2 and 3-4 would be represented by vertical lines on the TS diagram and would look more like the Carnot cycle.
The Rankine cycle shown here prevents the working fluid state from ending up in the superheated steam region after expansion in the steam turbine. In this case, the energy removed by the capacitors is reduced.
The actual steam power cycle differs from the ideal Rankine cycle due to irreversibilities caused by fluid friction and heat loss to the surroundings. The friction of the fluid causes pressure drops during the fluid circuit. In turn, the loss of heat reduces the net output of work and requires the addition of heat to the steam.
What is the Rankine cycle used for?
The cycle describes the operation of thermal machines in thermal power plants. The usual sources of heat for these plants are fossil fuels, solar energy or nuclear fuel.
Although a Rankine cycle can work with various substances, water is often used due to several favorable properties. These plants use water as the motor fluid, either in liquid form or in the form of steam or gas, with the so-called steam turbine.
The relatively low temperatures in the Rankine cycle mean that this cycle is used as a low temperature cycle in thermal power plants operating after a combined steam and gas cycle.
On the other hand, it is rapidly falling into disuse in the field of railway traction and marine propulsion, supplanted by the Diesel engine and the electric motor.