A heat exchanger is a technical device in which heat exchange occurs between two media with different temperatures.
A solar heat exchanger is a device designed specifically to do this task in a solar thermal system. Cold water - a heat transfer fluid - enters the solar collector, and solar radiation hits the collectors' surface area, heating the water flowing through them. This fluid is specifically treated to increase its energy efficiency and flows in a closed circuit, the primary circuit.
When the fluid is hot, it goes to the solar heat exchanger to transfer its thermal energy to the secondary circuit without mixing. The liquid of the secondary circuit is regular water that can be stored in an insulated tank. Sometimes, hot water is used to harness a solar water heating system or in a climatized swimming pool.
How does a heat exchanger work?
The way a heat exchanger works depends on the type of exchanger. However, the principle of operation is the same.
The exchanger has two inlets and two outlets through which two fluids enter and leave at different temperatures. Both fluids come into contact with a surface with a high heat transfer, each on the one hand, without mixing.
The hotter fluid gives up heat to the surface, raising the temperature and its internal energy. At the same time, the surface gives up this thermal energy to the cold fluid in the form of heat.
Energy-efficient heat exchangers are designed to reduce waste heat, focusing mainly on two points:
-
Reducing the surface area to reduce the contact with air flow.
-
Wrap with insulating materials to reduce the waste of heat.
What is the law of heat exchange?
The law of heat exchange is the set of laws of thermodynamics that regulate the transfer of heat between two elements:
-
Suppose we have two elements at different temperatures in contact with another part. Energy will pass from one to another until all three have the same temperature (first law of thermodynamics).
-
Heat can only flow from a hot element to a colder (second law of thermodynamics).
-
The heat transfer rate is proportional to the temperature and flow rate difference.
Types of heat exchangers
Depending on the design of the exchangers, the main types are:
1. Double tube heat exchanger (or "concentric tubes")
Fluids flow in two coaxial tubes, one inside and one outside. In this exchanger, the liquid with a higher temperature flows through the inner conduit, giving up heat to the fluid with a lower temperature that flows around it. They are also known as jacketed exchangers.
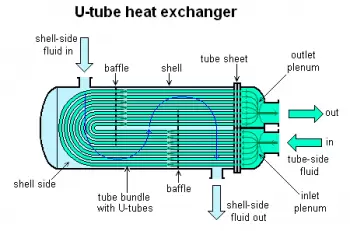
2. Shell and tube heat exchanger
In the shell and tube exchanger, one of the fluids passes through the interior and the other through the exterior of the tubes, in a specially designed chamber (shell or shell).
3. Helical heat exchanger
It consists of a helical or serpentine-shaped tube into which the process fluid flows, which is cooled through a film of water that drips from above.
4. Plate heat exchangers
Plate heat exchangers consist of a set of plates in which the fluids flow in alternating channels, against the current.
A particular case is the gasket plate heat exchanger. The plates have channels sealed with elastomeric gaskets in this exchanger type, and the flow flows into alternative media—the plate pack mounts between a frame and a bolt-on pressure plate.
5. Spiral exchanger
the two fluids pass to the opposite sides of a sheet, generally smooth, in single chambers of great length, wound in a spiral.
6. Exchanger for graphite or other material
The currents circulate in cylindrical holes, generally arranged orthogonally on the two sides; extended surface exchangers.
Other types of heat exchangers
There are other classifications of exchangers:
According to the flow direction
-
Parallel flow: hot and cold fluids flow in the same direction.
-
Counter flow: fluids enter at different ends and circulate in the same direction but in opposite directions. In this case, heat recovery is much more efficient than in parallel flow.
-
Cross flow: fluids flow through a bundle of tubes perpendicular to each other.
-
Single-pass: in this exchanger type, the fluid's heat only exchangest once. Sometimes the heat energy transfer needs to be increased, and two or more exchangers are combined in series.
-
Multiple Steps: Fluids exchange heat more times.
-
Regenerative and non-regenerative: whether the same fluid is used or not.
According to the type of contact
Depending on how the fluids interact, we distinguish the following heat exchangers:
-
Direct contact: the interfaces of the heat-exchanging fluids are in direct contact with each other.
-
Indirect contact: the fluid interfaces are separated by a surface that transmits heat.
Examples of use of heat exchangers
Heat exchangers are used in technological processes in the oil refining, petrochemical, chemical, nuclear power, refrigeration, gas and other industries.
In solar energy systems, the heat exchanger transfers the heat captured through solar radiation to another working fluid. Solar thermal energy can be used both to supply thermal energy in a heating system and solar thermal power plants.
Other examples of standard heat exchangers are the car radiator and the heater for domestic heating.