
The zeroth law of thermodynamics deals with the exchange of energy, mainly in the form of heat, between physical systems. This law establishes an essential concept: thermal equilibrium.
The formal statement of the zeroth law of thermodynamics is as follows:
If two systems A and B are in thermal equilibrium with a third system C, then A and B are in thermal equilibrium with each other.
This law implies that temperature is a fundamental property that can be measured consistently across different systems. By establishing that thermal equilibrium is a transitive condition, the zeroth law allows temperature to be defined as a property that can be equated across different systems, facilitating its measurement through a reference device (such as a thermometer).
Thermal equilibrium occurs when two systems in thermal contact no longer exchange energy in the form of heat. In other words, when there is no temperature difference between two systems, they reach thermal equilibrium. If a system A is in thermal equilibrium with another system B, and this in turn with a system C, then A and C will also be in thermal equilibrium.
Below are several examples that illustrate the zeroth law of thermodynamics in everyday and scientific situations:
1. Mercury thermometer
One of the most common examples of the zeroth law of thermodynamics is the use of a thermometer to measure the temperature of an object or environment. A traditional mercury thermometer consists of a glass tube with mercury inside, the volume of which varies depending on the temperature.
When the thermometer is placed in contact with an object (for example, a cup of hot water), the mercury inside the thermometer expands or contracts until the thermometer reaches thermal equilibrium with the object.
According to the zeroth law, if the thermometer and water are in thermal equilibrium, then the temperature of the water is the same as that of the thermometer, allowing for an accurate measurement.
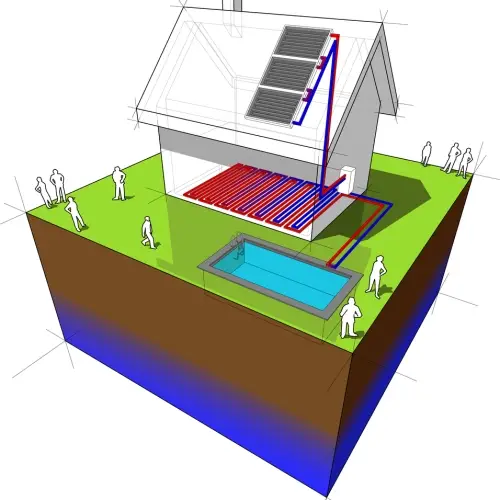
2. Solar thermal energy panels
 Solar thermal energy is an efficient method of capturing the sun's energy and converting it into heat, which can then be used to heat water or air, or even generate electricity.
Solar thermal energy is an efficient method of capturing the sun's energy and converting it into heat, which can then be used to heat water or air, or even generate electricity.
In solar thermal energy systems, such as solar hot water collectors, panels capture solar radiation and use it to heat a fluid, usually water or a specialized liquid.
Imagine a solar water heating system. Solar thermal panels (system "A") are exposed to solar radiation, which heats the fluid inside the collectors. This hot fluid transfers heat to cold water circulating in a storage tank (system "B").
Initially, the water in the tank is at a lower temperature than the fluid in the solar collectors. As heat is exchanged between the two, the water is progressively heated.
According to the zeroth law of thermodynamics, when the water in the tank reaches thermal equilibrium with the fluid heated by the sun, both will have the same temperature and heat exchange will stop.
3. Air conditioning system in a home
 Heating and air conditioning systems in homes also obey the zeroth law of thermodynamics.
Heating and air conditioning systems in homes also obey the zeroth law of thermodynamics.
Imagine that you set your home thermostat to a desired temperature, for example, 22°C. The heating or cooling system will activate to increase or decrease the air temperature until the temperature in all rooms reaches the value set on the thermostat.
In this case, the thermostat acts as the reference system "C", and the rooms in the house would be the systems "A" and "B". When all the rooms are in thermal equilibrium with the thermostat, that is, when they all reach the programmed temperature, they are also in thermal equilibrium with each other.
4. Cooling a drink in a refrigerator
Another everyday example is cooling a beverage in a refrigerator. Imagine that you put a can of soda at room temperature into a refrigerator, whose internal temperature is set to 5°C. The refrigerator acts as the cold system (system "C"), while the can of soda is system "A". Over time, the can transfers heat to the cold air in the refrigerator until they both reach thermal equilibrium, i.e. the same temperature.
According to the zeroth law, once the beverage has reached 5°C, it will be in thermal equilibrium with the refrigerator and no further heat transfer will occur. At this point, the temperature of the can and the refrigerator are equal.
5. Internal combustion engine
 In the context of engineering and technology, the zeroth law also applies to the operation of an internal combustion engine. During engine operation, different parts of the system, such as the engine block and radiator, must manage large amounts of heat.
In the context of engineering and technology, the zeroth law also applies to the operation of an internal combustion engine. During engine operation, different parts of the system, such as the engine block and radiator, must manage large amounts of heat.
The radiator, for example, is responsible for cooling the engine to prevent overheating. The coolant inside the radiator exchanges heat with the hot engine until both reach thermal equilibrium. In this way, the engine temperature is kept within a safe range for its operation.
6. Industrial cooling processes
In industry, especially in the manufacturing of chemicals or food, temperature control ensures the quality and safety of products.
For example, in the pasteurization of milk, the milk is heated to a specific temperature to kill bacteria and then rapidly cooled to a safe temperature for consumption.
During the cooling process, hot milk comes into contact with cold metal plates. As the milk transfers heat to the plates, it cools and reaches thermal equilibrium with the plates.
Here, the zeroth law of thermodynamics ensures that at the end of the process, the milk and the plates will have the same temperature, allowing precise control of the process conditions.
7. Bain-marie in the kitchen
 The bain-marie is a culinary technique used to slowly heat food without the risk of burning or overheating it.
The bain-marie is a culinary technique used to slowly heat food without the risk of burning or overheating it.
The method consists of placing a container with food on top of another containing hot water. As the water transfers heat to the upper container, the food reaches thermal equilibrium with the water, allowing it to remain at a constant temperature without exceeding it.
In this case, system "C" is the hot water, which is in thermal equilibrium with the container "A" containing the food. The zeroth law of thermodynamics states that if the water and the container are in thermal equilibrium, then the food will also be in thermal equilibrium with the water, maintaining controlled and uniform cooking.
8. Aquarium thermostat
Temperature control in aquariums is essential for the health of the fish and plants that live there.
A thermostat submerged inside the aquarium is used to measure and regulate the water temperature. The thermostat is in direct contact with the water and adjusts heating or cooling to maintain the proper temperature.
According to the zeroth law, the thermostat reaches thermal equilibrium with the aquarium water. When the thermostat temperature stabilizes, we can infer that all the water in the aquarium is at the same temperature as that displayed on the device, allowing the animals and plants to remain in a controlled and healthy environment.
9. The human body and the external climate
 The human body is highly sensitive to temperature changes and uses mechanisms such as sweating or shivering to regulate its internal temperature. Imagine a person entering a cold room. Initially, their body will be at a higher temperature than the air in the room, causing them to start losing heat to their surroundings.
The human body is highly sensitive to temperature changes and uses mechanisms such as sweating or shivering to regulate its internal temperature. Imagine a person entering a cold room. Initially, their body will be at a higher temperature than the air in the room, causing them to start losing heat to their surroundings.
Over time, the body and the air in the room will reach thermal equilibrium, at which point the person will no longer lose heat to the environment.
This phenomenon can be observed when we stay in a cold place and we begin to feel that the sensation of cold stabilizes, since we have reached thermal equilibrium with the environment.
10. Heat exchangers in power plants
 In power plants such as nuclear power plants or conventional power plants, heat exchangers are devices that transfer thermal energy between fluids.
In power plants such as nuclear power plants or conventional power plants, heat exchangers are devices that transfer thermal energy between fluids.
A typical example is the use of a hot fluid that transfers its heat to a cold fluid without the two coming into direct contact. This is key in industrial processes to recover energy and increase efficiency.
The zeroth law applies in this case when the two fluids reach thermal equilibrium inside the exchanger. Once both fluids have equalized their temperatures, the heat exchange ceases. This thermal equilibrium is crucial to ensure that power generation and distribution systems operate efficiently and safely.
11. Electric coffee maker and coffee cup
Finally, we propose the example of an electric coffee maker and a cup of coffee.
Imagine you are making a cup of coffee using an electric coffee maker. When you pour the hot coffee into a ceramic cup at room temperature, there will initially be a heat exchange between the coffee and the cup. The hot coffee transfers heat to the cooler cup, and eventually the two will reach thermal equilibrium.
The zeroth law is at work here because once the coffee and the cup have stopped exchanging heat, they are in thermal equilibrium, meaning they both have the same temperature. If you had a thermometer, you could measure this final temperature, knowing that both the cup and the coffee are in thermal equilibrium.