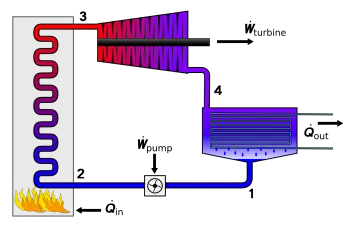
A thermodynamic cycle consists of thermodynamic processes that involve the transfer of heat energy. This sequence of thermodynamic processes aims to obtain work from two heat sources at different temperatures, or inversely, to produce, through the contribution of work, the passage of heat from the source of lower temperature to higher temperature.
Obtaining work from two heat sources at different temperatures is used to produce motion—for example, the drive of turbines for the generation of electrical energy.
In a reverse thermodynamic cycle, we get heat by exerting some mechanical work. Then, external work is added to the process to cause heat transfer to occur from the cooler source to the hotter source, in reverse of how it would tend to happen naturally. This arrangement is used in air conditioning and refrigeration cycles.
Types of thermodynamic cycles
We can classify thermodynamic cycles according to their reversibility:
Reversible Cycle
We define a reversible cycle as a cycle in which we do not lose energy for any cause, such as radiation, conduction, or friction. Therefore, in a reversible process, it is possible to restore it to its original state when it is finished.
If there is no energy exchange out the system while varying pressure, temperature, or other thermodynamic properties, we can repeatedly repeat the process on both sides.
For example, if you take some water at a constant volume isolated from its surroundings and start exerting pressure on it, it will change into steam. Of course, with varying pressure and temperature, we can change the initial state. However, if we return the pressure to its initial state, steam will return to its liquid form.
Irreversible Cycle
In thermodynamics, an irreversible cycle is a process that, like entropy, is not reversible in time. From this thermodynamic perspective, all-natural processes are irreversible.
The German physicist Rudolf Clausius, in the 1850s, was the first who quantified the phenomenon of irreversibility in nature, and he did so through the introduction of the concept of entropy.
Thermodynamic cycle’s performance
The performance is defined as the work obtained divided by the heat released in the thermodynamic process, in the same complete cycle time if the process is continuous. This parameter is different according to the multiple thermodynamic cycles, but the Carnot factor or yield limits it.
Efficiency is the main parameter that characterizes a thermodynamic cycle.
Examples of thermodynamic cycles
There are many types of thermodynamic cycles. Here are some of the prominent examples:
1. Carnot cycle
The Carnot cycle is a theoretical cycle designed to compare the thermal efficiency of heat engines. It is a reversible cycle carried out by a "Carnot engine" connected to two sources of different temperatures. It uses an ideal gas as a working agent through whose transformations mechanical work is obtained.
2. Organic Rankine Cycle
The Organic Rankine Cycle is a predictive model of the operation of a gas turbine system. This model uses a high molecular weight organic working fluid with a liquid-to-vapor phase change, or boiling point, at a lower temperature than the water-to-vapor phase change.
3. Diesel Cycle
The diesel cycle is one of the most used cycles in the thermal engines of automobiles.
In diesel engines, the movement is produced by the self-ignition of the fuel due to high temperatures after the fuel compression.
Four processes occur during the diesel cycle: two isentropic processes alternated with an isochoric process and an isobaric process.
4. Otto Cycle
The Otto cycle is the thermodynamic cycle applied to internal combustion engines with ignition caused by an electric spark (gasoline engines, ethanol, petroleum gases, or other highly volatile and flammable substances). All the heat is supplied at a constant volume in an Otto cycle.
Nicolaus Otto invented this cycle in 1876, which was just a theoretical approximation.
5. Stirling Cycle
The Stirling cycle is a thermodynamic cycle expressing the engines of the Stirling principle.
The Stirling cycle is considered a reversible cycle. If some mechanical energy is added to the cycle, it will act as a heat pump for heating or cooling and even deep or extreme cooling.
It is also a closed cycle in which the fluid that flows inside it never leaves the process.
6. Bryton Cycle
The Brayton cycle, also known as the Joule cycle or the Froude cycle, is a thermodynamic cycle. It consists of an adiabatic compression stage, an isobaric heating stage, and an adiabatic expansion of a compressible thermodynamic fluid.
It is one of the most widely applied thermodynamic cycles, as it is the basis of the gas turbine engine.
The product of the cycle can range from mechanical work used for the production of electricity in natural gas burners or some other use –case of the electricity generation industries and some land or marine engines, respectively–, to the generation of a thrust in a jet engine.