Among the fundamental laws that govern thermodynamics, the zeroth law occupies a crucial place, since it establishes the fundamental concepts of temperature and thermal equilibrium.
The zeroth law of thermodynamics states that "if two systems are in thermal equilibrium with a third system, they are also in equilibrium with each other." It is also known as the zero principle of thermodynamics.
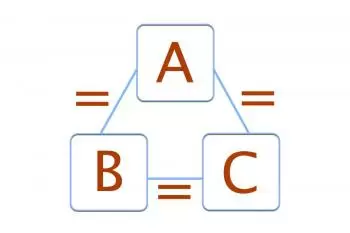
If A is in equilibrium with B and A is also in thermal equilibrium with a third body C, we can conclude that B is in thermal equilibrium with C.
Two bodies are in thermal equilibrium when, upon contact, their state variables do not change. If they are not in thermal equilibrium, they will undergo an exchange of heat or energy and reach thermal equilibrium.
The zeroth law was the last of the laws of thermodynamics to be introduced. To obtain a logical structure, it was necessary to place it before the other laws.
Formula of the 0th law of thermodynamics
Principle 0 of thermodynamics, if the three systems are in thermal equilibrium, the following formula must be completed:
TA = TB = TC
Where TA , TB and TC are the temperatures of the three systems.
Origin and history of the zero law
The zeroth law of thermodynamics was formulated as an answer to questions about measuring and comparing temperatures. Before their establishment, temperature scales were arbitrary and depended on specific measurement methods. The need for a more solid basis for comparing temperatures was a hot topic in the 19th century scientific community.
It was the German physicist Gustav H. Johannsen who first formulated the central idea of the zeroth law in the 1860s, but it was not until the 1930s that it was consolidated and incorporated as a fundamental law of thermodynamics.
The zeroth law is so called because it provides the basis for the construction of temperature scales and establishes a fundamental principle for measuring temperature.
Examples of the zeroth law
To understand how the zeroth law of thermodynamics works, we propose three examples:
Ice and water
We put a piece of ice and a glass full of water at room temperature. In this experiment three elements appear: ice, water and the glass.
Initially the three will exchange heat and energy until reaching thermal equilibrium. After a while, the three elements will reach the same temperature
Cups of coffee
Let's imagine two cups of coffee, one hot and one cold. When they come into contact, heat flows from the hot cup to the cold cup until both reach the same temperature and are in thermal equilibrium.
This is a practical example of the zero principle in action: the two systems are in thermal equilibrium when there is no longer a net transfer of heat between them.
Thermometer example
Another example that will help better understand the concept of thermal equilibrium is that of a thermometer.
Consider a body B that consists of a tube with a capillary containing mercury. Their height levels above the capillary represent different temperatures.
Now, consider a body A, for example the human body. If you bring the thermometer close to your body and leave it for long enough, the thermometer will reach the value corresponding to your body temperature.
The thermometer and the human body will be in thermal equilibrium with each other. Therefore, both elements reach the same temperature.